Gore Medical
Gore Medical Products Division engineers devices that treat a range of cardiovascular and other health conditions. With more than 40 million medical devices implanted over the course of more than 40 years, Gore builds on its legacy of improving patient outcomes through research, education and quality initiatives. Product performance, ease of use, and quality of service provide sustainable cost savings for physicians, hospitals and insurers. Gore is joined in service with clinicians, and through this collaboration we are improving lives.
Case Discussion: Applying Patent Foramen Ovale and Secondary Stroke Prevention Guideline in the Real World
By
Gore Medical
FEATURING
John J. Volpi
By
Gore Medical
FEATURING
John J. Volpi
The AAN released an updated practice advisory in April 2020 on “Patent Foramen Ovale and secondary stroke prevention” with a recommendation for PFO closure in select patients.
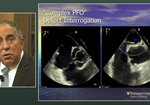
John J. Volpi, M.D., a Vascular Neurologist at Houston Methodist and director of the Houston Methodist Hospital Comprehensive Stroke Center shares a case discussion using the guideline in the real world.
John J. Volpi, M.D., a Vascular Neurologist at Houston Methodist and director of the Houston Methodist Hospital Comprehensive Stroke Center shares a case discussion using the guideline in the real world.
Login to view comments.
Click here to Login